Nanomaterials for energy
Nanomaterials play an important role in the development of new technologies for energy storage and conservation. The properties of materials are highly dependent on both their atomic arrangement and nanostructure. It is therefore crucial to be able to precisely control these particle characteristics during synthesis.
We use especially solution-state synthesis to form novel nanomaterials with tailormade characteristics. We are particularly interested in ultrasmall nanoparticles and clusters with dimensions below 5 nm, where the atomic structure and material properties fundamentally differ from the bulk.
X-ray and neutron scattering for nanomaterial characterization
The development of materials for energy applications builds on an intricate understanding of the relation between material structure and properties. Only by knowing the atomic arrangement can the mechanisms responsible for material properties be elucidated and new materials developed.
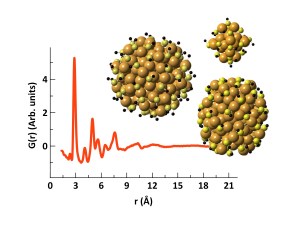
We use X-ray and neutron scattering to study the atomic structure of materials. By combining traditional crystallographic methods with total scattering techniques, we are able to elucidate the structures even in ultrasmall nanoparticles and thereby pinpoint how they structurally differ from the bulk.
For examples of our research on nanostructure analysis using total scattering, see. e.g.:
Understanding the formation of nanoparticles – watching materials form with X-rays
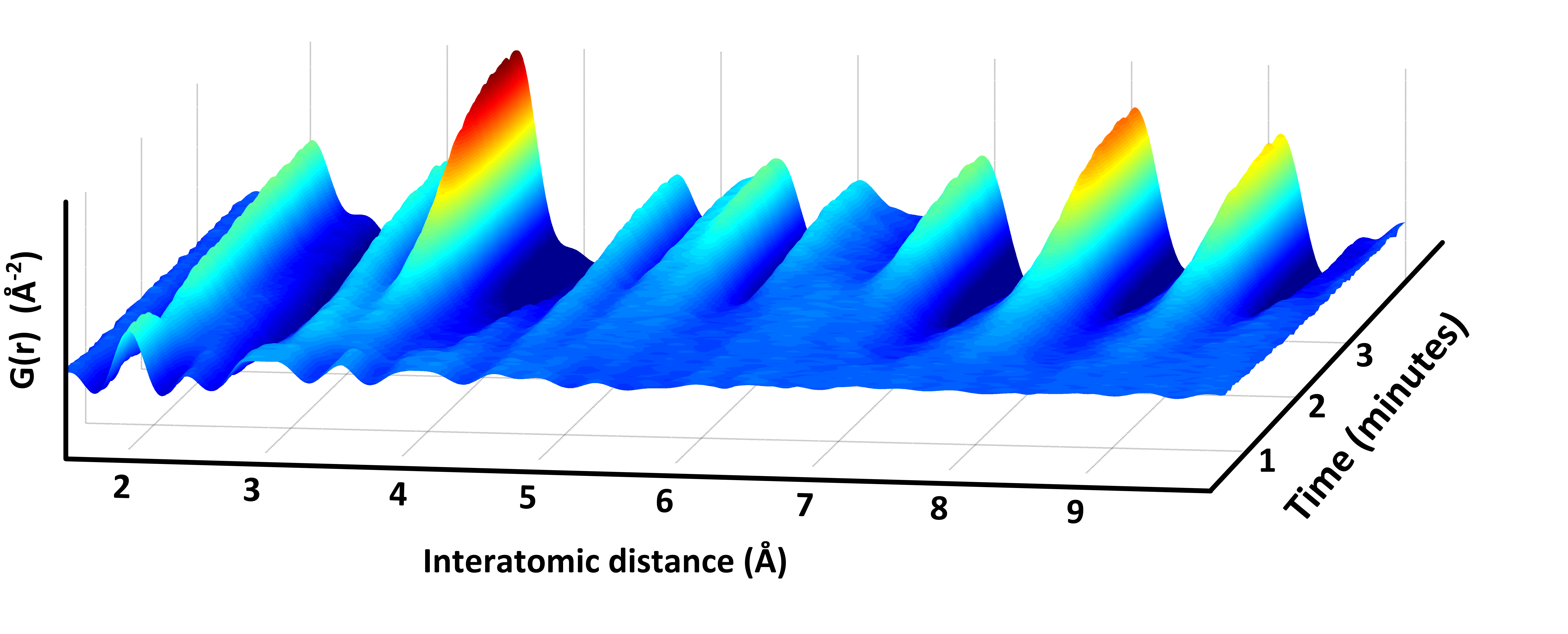
X-ray total scattering allows structural information to be obtained from both amorphous and crystalline samples; liquids as well as solids. By using X-ray total scattering in situ during nanoparticle formation, i.e., by measuring time-resolved X-ray data as the synthesis takes place, we are able to follow the structural transformation that occurs as atoms arrange into ordered nanoparticles. This gives us new insight into reaction mechanisms, taking us one step closer to ‘materials by design’.
For examples of our in situ X-ray total scattering studies, see e.g.:
Analysing scattering data using Machine Learning based methods
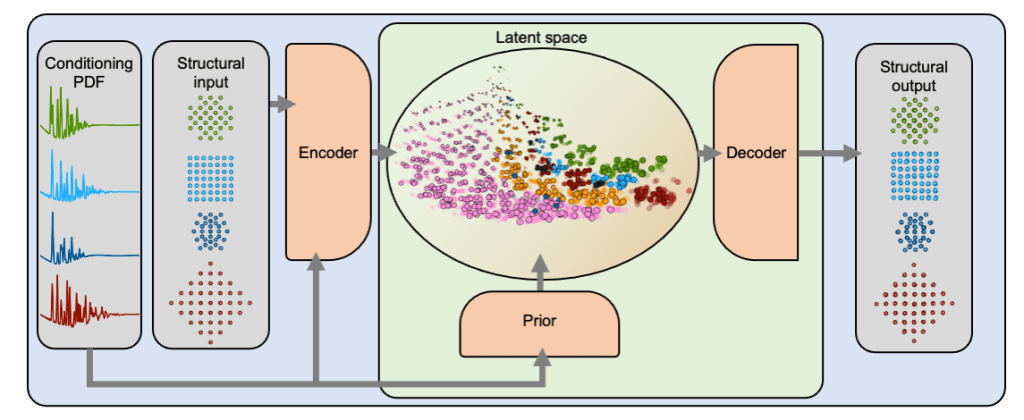
While scattering techniques have been used to analyse the atomic structure of materials for a century, it is still not straightforward to determine the structure of, e.g., nanomaterials based on a given scattering pattern. We can analytically calculate the scattering pattern from a material but not vice versa. This is called the inverse problem.
We work on solving this inverse problem by developing machine learning based methods and training them on large databases consisting of structures and their calculated scattering patterns.
For examples of our research on machine learning based scattering analysis, see e.g.:
Kjær & Anker et al.: DeepStruc: Towards structure solution from pair distribution function data using deep generative models, Digital Discovery, 2, 69-80, 2023
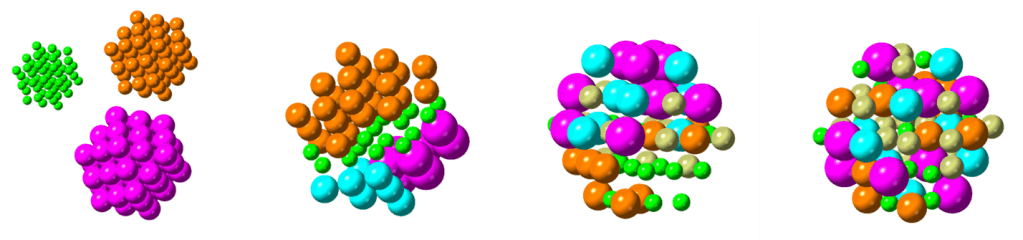
Center of High Entropy Alloy Catalysis (CHEAC)
As part of the Center of Excellence, Center for High Entropy Alloy Catalysis (CHEAC), we aim to synthesize and structurally characterize novel high entropy alloys and high entropy oxides. High entropy alloys and oxides are new material classes consisting of at least five elements that are randomly mixed with each other in solid solutions. This element mixing strategy opens up a vast compositional space that is still to be explored. The surface area of these random element particles can have millions of different local structures, which can act as active catalytic centers. This approach allows for a new, statistical way of designing new catalyst materials, which our colleagues in CHEAC (Jan Rossmeisl’s group at UCPH) explore theoretically. The electrocatalytic properties of the materials are tested in CHEAC as well, in collaboration with the groups of María Escudero-Escribano (UCPH) and Matthias Arenz (University of Bern).
In the nanostructure group, we focus on developing synthesis routes for nanoparticles of these new classes of materials. We explore their characterization with X-ray and neutron based techniques and study the formation mechanisms to understand what determines the formation of mixed or non-mixed particles.
For examples of the research performed at CHEAC, see e.g.: